Work programme
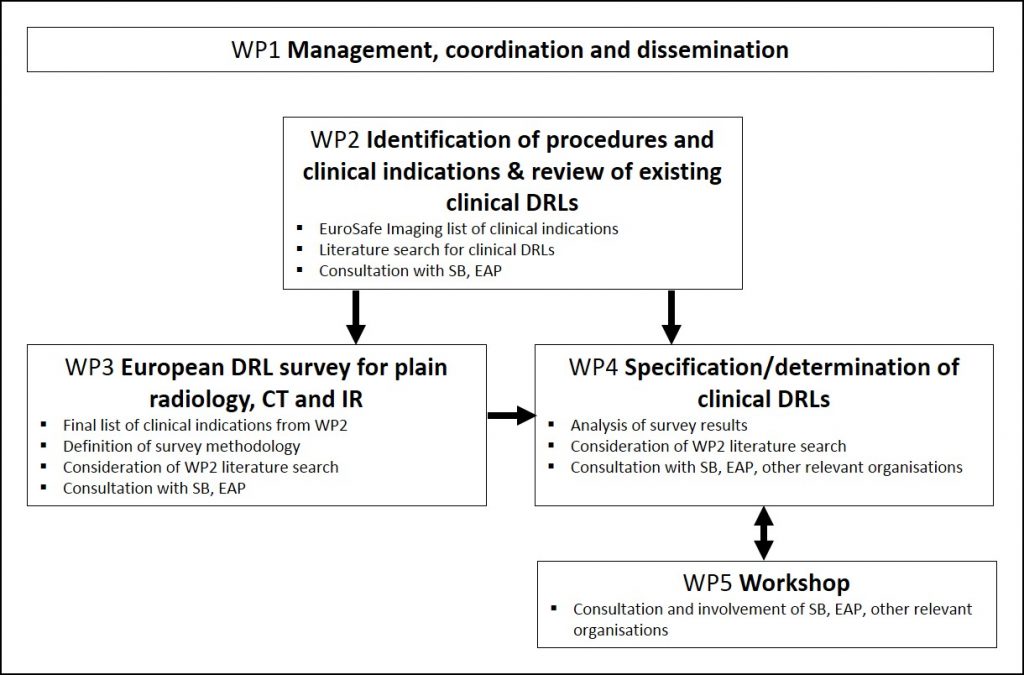
The EUCLID project is divided into 5 interrelated work packages (WPs), which cover specific tasks leading to the common objective to carry out a European study on clinical DRLs for x-ray medical imaging:
- Management and general coordination of the project, as well as dissemination (WP 1);
- Identification of procedures and clinical indications for which DLRs will be established, as well as review of existing DRLs (WP 2);
- Implementation of a European DRL survey for plain radiology, CT and IR following a predefined methodology (WP 3);
- Specification/determination of up-to-date clinical DRLs for Europe for the protocols/imaging tasks identified under WP 2 and stakeholder consultation/validation of the DRLs (WP 4);
- Organisation of a workshop to disseminate and discuss the results of the project with Member States and relevant national, European and international stakeholders and to identify the need of further national and local actions on establishing, updating and using DRLs (WP 5).
The below figure shows the work package structure and interrelation of the WPs.